Skin Models
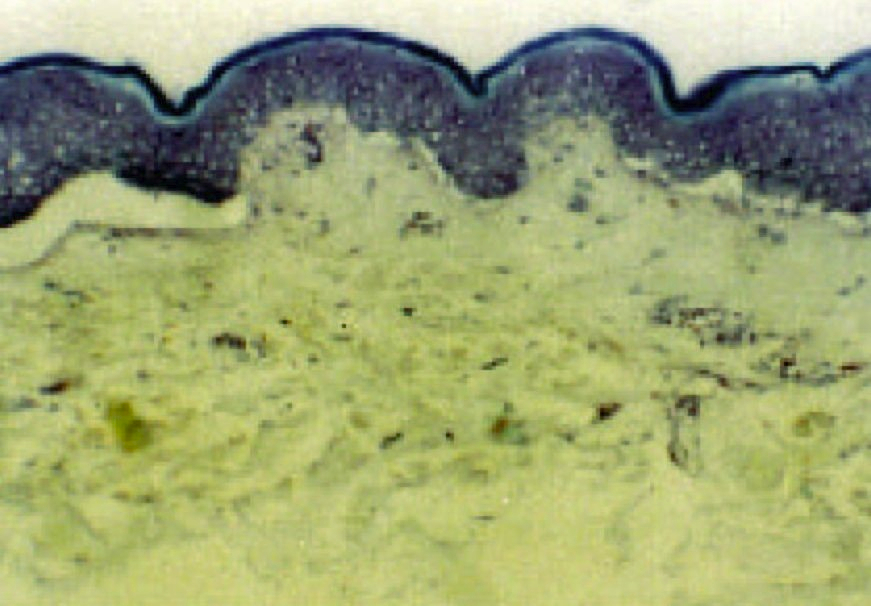
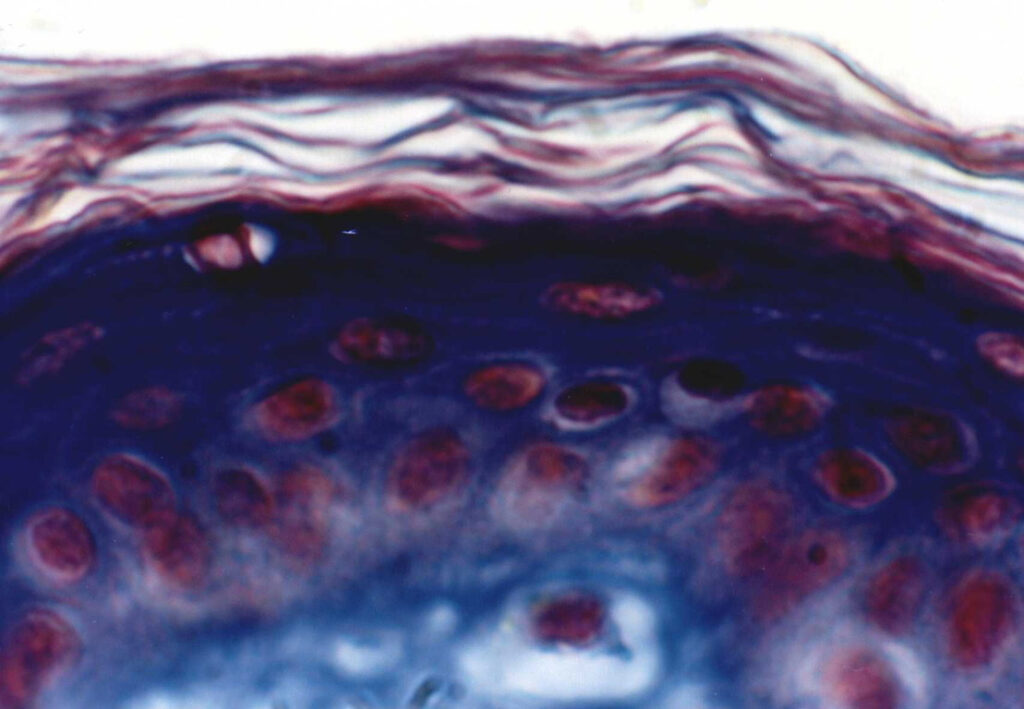
In recent years there has been increasing interest in the dermal application of drugs due to the simplicity of this type of delivery system. Despite this positive trend, costly and time-consuming clinical studies must still be performed in order to determine dermatopharmacokinetic parameters. Modern in vitro studies complement conventional clinical research and now make a significant contribution to the characterization of topically applied drug formulations.
Our prepared Human Skin can be used for experiments following the guidelines:
- OECD 428
- OECD 431
- OECD 435
- OECD 439
- ISO 10993-10
Drugs that are topically applied but that act only in deeper lying skin tissue can now be studied using novel test models and modern analytical techniques without the need for extensive and costly clinical investigations. For any new drug preparation it is nowpossible to determine in vitro whether the pharmaceutical active ingredient is able to penetrate the skin “barrier”.
in vitro studies also offer an interesting alternative when optimizing the composition of a formulation to achieve the required drug uptake across the skin, when investigating the interaction of chemicals with the skin, or when developing cosmetic products.
For detailed information please visit our Human Skin Bank or contact us directly.